Introduction
Multinational clinical studies have faced challenges with patient retention for an extended period of time. Despite the importance of these studies in gathering data from different patient populations, these studies have some challenges such as transport issues, culture, language, barriers, and differences in regulations. Studies with high attrition rates are costly because of the extended timelines and also result in lower quality data.
Retention Challenges in Multinational Studies
Multinational Clinical studies and retention of patients have no simple solutions and many barriers, such as:
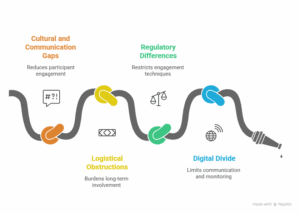
- Gaps in culture and communication – Participants from various locations in the world may lack engagement and may have different views on the requirements of the study.
- Logistical Obstructions – Long-term involvement in the study may be difficult because of the needed time for various appointments as well as the distance that needs to be traveled.
- Regulatory Differences – Rules in different countries may restrict the use of common engagement techniques.
- Digital Divide – The lack of technology in some areas can restrict communication as well as the ability to monitor patients.
These obstacles illustrate that the use of patient-centered solutions that are flexible and innovative are required.
ProCTTH’s Role and the Emerging Importance of Digital Transformation
ProCTTH has been pioneering the redefining retention strategies for global clinical trials. With the use of digital tools, automated workflows, and proactive communication tools, ProCTTH keeps patients actively engaged and supports every step of the study. The use of digital transformation into clinical trials has crossed from being optional to a requirement.
Key benefits include:
- Improved retention through predictive, proactive analytics to mitigate dropout risk.
- Telehealth, wearables, and virtual visit technologies for patient-centered remote care.
- Global consistency of operational workflows across regional boundaries.
ProCTTH’s digital-first approach, in a fast-growing and dynamic research environment, has gone beyond retention challenge to redefine industry standards in agility, compliance, patient experience, and overall efficiency in cross-border clinical studies.
What Are Patient-Centric Clinical Trial Apps?
Centrally focused trail apps. or patient centric clinical trial apps, are designed to increase engagement and patient ease during a clinical trial. Unlike previous trial apps, clinical trial apps aimed to streamline the process to make participation user-friendly. Sponsoring participants, trials, collection data, and the patient’s experience should all decrease time, increase ease, and enhance satisfaction to ultimately increase participation, retention, and precision.
Core Features of Patient-Centric Trial Apps
Ease of Data Entry
User-friendly interfaces make it easy for the patient to log their health, symptoms and activities. Patient burden is minimized and compliance is increased due to a simple design and lower clicks per task.
Reminders and Notifications
Missed data points are minimized due to manual reminders alerting the patient to take their medication, complete questionnaires, or follow-up visits.
Feedback and Insight
Patients wait to give a portion of their involvement and feel contribution. Many apps offer summaries or feedback instantly, emphasizing the importance of engagement. Such transparency fosters trust.
Multilingual Support
Trials oftentimes are meant for a specific group of people, however, patient user-friendly apps increase participation, by removing the barriers of language.
Why They Matter
Overall experience is one of the important aspects and patient-centric trial apps intersperse the life of a participant and the intricacies of a clinical trial. They boost patient retention and satisfaction rates while closing the feedback loop – supplementing trial sponsors with valuable and quality real world data.
The Problem: Retention in Multinational Clinical Trials
Retention is still one of the biggest challenges in multinational clinical trials. Global data shows that 20–30% of patients drop out of clinical trials. In particular therapeutic areas or in the case of digital/app-based interventions, attrition rises to over 40%. This phenomenon of dropout is not only bad for the power of the study, but also delays timelines, inflates cost, and undermines the confidence of regulators on the study outcomes.
The primary challenges are mostly the same everywhere, the burden of time and travel, complicated protocols, adverse events and other socio-economic challenges, and misunderstanding of the trial procedures. The challenges associated with multicountry and multicultural clinical trials are further complicated by other problems. Inadequate translation, or translation of poor quality, of consent forms and study materials creates a communication barrier.
Cultural differences shape trust, health beliefs and the willingness to continue participation. The retention strategies themselves, as reimbursement or follow up, are complicated by divergent regulatory and ethical standards. The retention is also complicated by infrastructural gaps, inconsistent standards of care, and the phenomenon of patient migration over different regions. In the end, retention is regionally refracted in a unique manner. A universal approach is ineffective. Therefore, retention strategies to be employed by sponsors or Contract Research Organizations, have to be direct, cross culturally sensitive and patient centered.
These approaches include, but are not limited to, flexible follow up procedures, simplification of complicated study protocols, reimbursement of travel and other associated costs, and the employment of local investigators as well as patient liaisons to help foster trust.In order to measure retention by site and minimize dropout during clinical trials, region specific proactive planning needs to be established, coupled with ongoing monitoring and retention strategies.
How ProCTTH Keeps People Involved In Clinical Trials
Retention of patients is one of the most problematic aspects of clinical studies. While the retention of patients increases a study’s data integrity, the dropouts stalled the streamlining of the study which studies tend to prioritize. ProCTTH platforms patients digitally during studies, enabling them to communicate and engage with site staff throughout the study.
Characteristics of the platform
- Remote patient monitoring – Patients can share any and all updates on their health from home, making them less inclined to visit the sites and more likely to engage.
- Flexible Scheduling – Patients do not attend any appointment with a rigid mindset. They understand that meetings can happen.
- Real-time engagement – Patients can stay on the study with the platform assurance that data and reminders on missed study activities will be collaborated.
- eConsent – Speeds up the enrollment processes by making them clearer and easily digestible for the patients.
- TeleHealth – Encourages patients, particularly stay-at-home patients, to converse without the hassle of relocating thus promoting a greater degree of accessibility.
- Personalized Direct Communication – Target people and make them feel important through the use of Direct-to- Speech patient centric ecosystem experiences. Patients are brought closer with the study and do not do the procedures formlessly, making them feel appreciated.
The Role of Technology on the Changing Clinical Trials
Patient centric trial app with the help of technology. Using decentralized clinical trials platforms helps alleviate the burden of physical visits to a clinic or hospital for a trial. Patient portals allow participants to have more control over the process. These portals provide transparency regarding the trial and allow participants to access real-time schedules and updates.
The use of gamification in patient-centric trial app provides a more user-friendly, motivating experience for trial participants. These participants are rewarded as they achieve various milestones through the software. AI-enabled are available as patient support and provide real-time reminders and customized support for participants. These technologies help in retention and minimizing dropout rates. Clinical Trials are also more efficient, with improved patient retention rates. These technologies are estimated to improve retention rates by 30%.
Conclusion
Retention in multinational clinical trials has proved to be one of the most intricate issues, mired in culture and geography, as well as regulations and technology. In the past, this was a major roadblock in the industry, unwilling to invest in patient-centric solutions. Today, the rise of digital-first solutions enable patient-engagement in cuffs, where ProCTTH is a prime example. Remote-centric technologies, eConsent, Telehealth, participant engagement, remote monitoring, and multilingual technology all help build trust and lessen participant drop-out rates.
Patient centric clinical trials apps that help the participant integrate better into the trial also enhance the trial experience by increasing the level of visibility, personalization, and reduction in accessibility barriers to trial-related information. In this era of globalized trials, it is the sponsors and CROs that are willing to be innovative and flexible, as well as culturally aware that will be positioned best to boost retention rates, data, and time efficiency. In this industry, patient commitment and defined success in multinational clinical research is something that can only be achieved if patient retention is worked on and this will be only achieved through digital transformation.
FAQ’s
1) What is another name for patient centric approach?
Answer: Another name for a patient centric clinical trials approach is a patient-centered (or patient-focused) clinical trials approach.
2) What are the four principles of patient centered care?
Answer: Listening to a clinical trial participant’s thoughts is an effective way to encourage them to openly share their experiences and perspectives. This approach provides support while motivating participants to take an active role in their journey, fostering greater engagement and adherence throughout the trial.
3) What are the benefits of patient centricity?
Answer: Patient centric clinical trials focuses on understanding and addressing participants’ needs and experiences. Key outcomes such as improved retention and adherence not only enhance trial efficiency but also lead to better health results. By prioritizing transparency and trust, patient centric clinical trials approaches encourage diverse participation and ensure every patient feels valued. Incorporating patient feedback drives meaningful innovations, enriches the trial experience, and ultimately benefits both patients and the advancement of research.
4) What is the patient centered method?
Answer: A patient-centered approach in clinical trials means that your needs, wishes, and concerns are always the priority. In this method, you play an active role in your treatment and decisions are made together with your doctors. There are open discussions about your care, and your physical, psychological, and social well-being are all taken into account. This approach makes participation easier by offering flexible scheduling, remote monitoring, and clear, supportive communication—helping you feel more comfortable and engaged throughout the trial.

